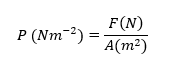Temperature ? Zeroth Law of Thermodynamic ? How do you visualize the temperature is ? Temperature is assosiated with the translation, rotation, vibration, and electronic states of the atoms and molecules that comprises matter. The more active the atoms and molecules the higher the temperature. It is like … mmm when you’re more active, like when you’re running, you feel like your body is warmer right ? you know that your body temperature is higher than before, this is happen because the atoms and molecules that comprises your body is more active. That is, when a object is brought into contact with another body that is at a different temperature, heat is transferred from the body at higher temperature to the one at lower temperature until both bodies attain the same temperature . At that point, the heat transfer stops, and the two bodies are said to have reached thermal equilibrium . The equality of temperature is the only requirement for thermal equilib...